The Future of Pharmaceutical Manufacturing Needs More than Capacity.
It Needs Control, Resilience, and Technology.
We deliver custom manufacturing solutions powered by dual R&D engines and geographically independent supply chains across China and India.
By combining proprietary continuous flow chemistry, micro-reactor technology, and AI-enabled process design, we help pharmaceutical innovators move faster — with better cost efficiency, higher quality, and stronger supply security.
With more than 25 years of experience supporting both global pharmaceutical leaders and clinical-stage companies, we specialize in advanced intermediates and regulatory starting materials for next-generation APIs.
Two Supply Chains. One Standard of Excellence.
Global supply resilience is no longer optional.
Our dual-platform model enables fully independent development and manufacturing pathways in both China and India — providing customers with flexibility, redundancy, and geopolitical risk mitigation.
China Platform
Proven Delivery at Scale
Our Chongqing-based R&D center drives process innovation for key building blocks used in clinical-stage and commercial APIs.
1,100 sqm research facility
20+ years of delivery experience
Integrated process, analytical, EHS, and QA teams
Technologies developed in-house are transferred to our network of six long-term manufacturing partners — each with extensive regulatory audit histories including:
FDA | EDQM | PMDA | MFDS
India Platform
A Fully Independent Supply Chain
Our India ecosystem offers true China-independent sourcing and production.
Through our strategic partnership with Sravathi Advanced Process Technologies*, we provide:
Locally sourced starting materials, reagents, and solvents
Backward integration capabilities
Advanced flow chemistry expertise
Founded by former GE and SABIC scientists, Sravathi brings:
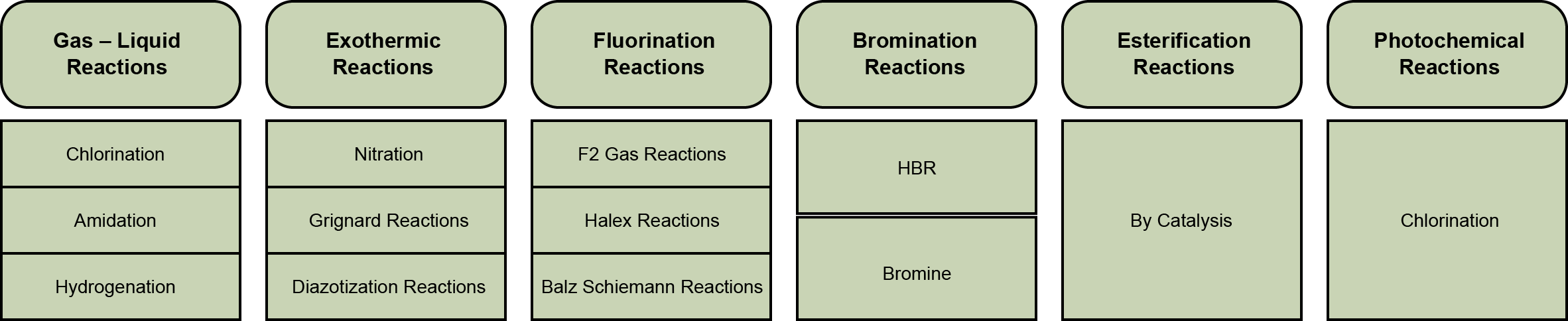
Validated micro-reaction expertise (nitration, fluorination, hydrogenation & more)
90 dedicated R&D scientists
AI-driven route optimization
*Ash is a 22% strategic equity shareholder.
Patented Microreactors
Validated Reactions via Proprietary Micro-reactors
Continuous Flow: A Structural Advantage
Traditional batch chemistry was built for yesterday’s challenges.
Our proprietary continuous flow and micro-reactor technologies deliver meaningful performance improvements:
20–30% lower energy consumption
Up to 94% reduction in residence time
Reduced effluent generation
Improved impurity profiles
Enhanced operator safety
Higher yields through tighter reaction control
The result: better economics and safer, more sustainable processes.
Next-Generation Peptide Manufacturing
Our process flow platform extends into peptide synthesis — unlocking new efficiency and scalability.
Through automated closed-loop flow systems and backward integration into protected amino acids, we enable:
Precise sequence construction
Improved process control
Reduced solvent use
Enhanced reproducibility
We have successfully synthesized peptides exceeding 40 amino acids using this approach.
This represents a potential step-change in peptide manufacturing.
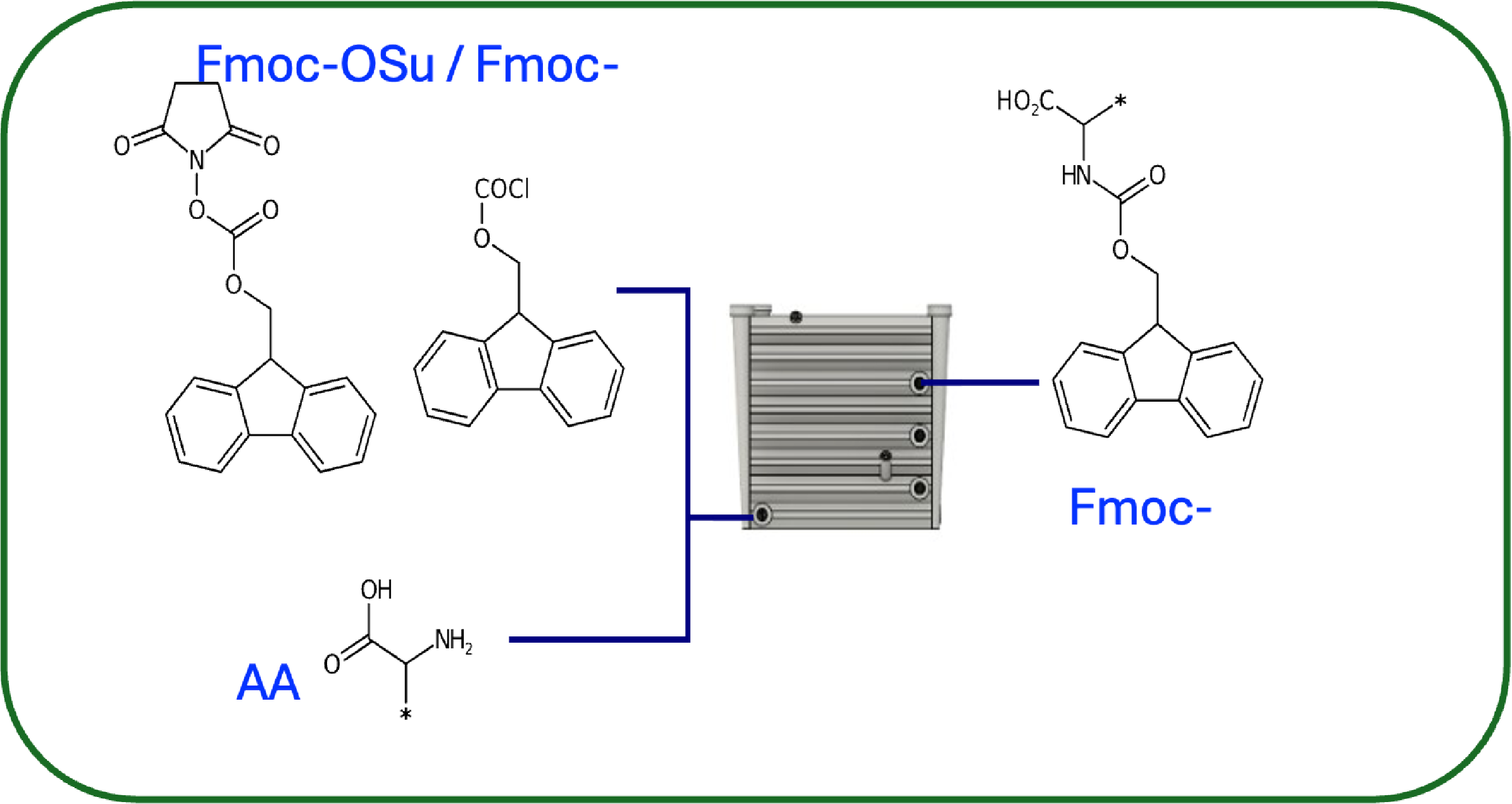
Backwards Integrated to create Protected Amino Acids
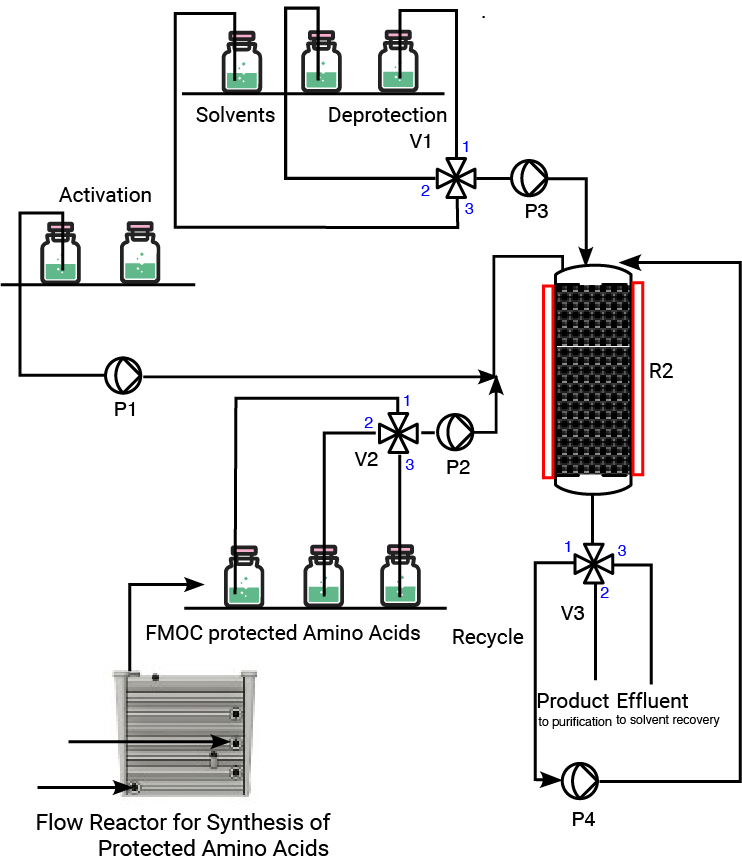
Automated Closed Process Flow System
Protected Amino Acid Synthesis
First protected Amino Acid (AA1) is grafted onto resin and placed into R2
Deprotection reagent (V1) travels into R2 and cleaves FMOC from AA1
Washing Solvents sent into R2 and effluent (FMOC, solvents) are flushed and separated through V3
FMOC-AA2 is loaded (V2) and combined with Activation agents into R1 for pre-activation; activated FMOC-AA2 sent into R2 to join AA1
Deprotection reagent (V1) travels into R2 and cleaves FMOC from AA2
AAs are continued to be added in this manner until completion of the sequence
Have successfully sequenced 40+ AA peptide to date
A Potential Game Changer for Peptide Synthesis
Why Us
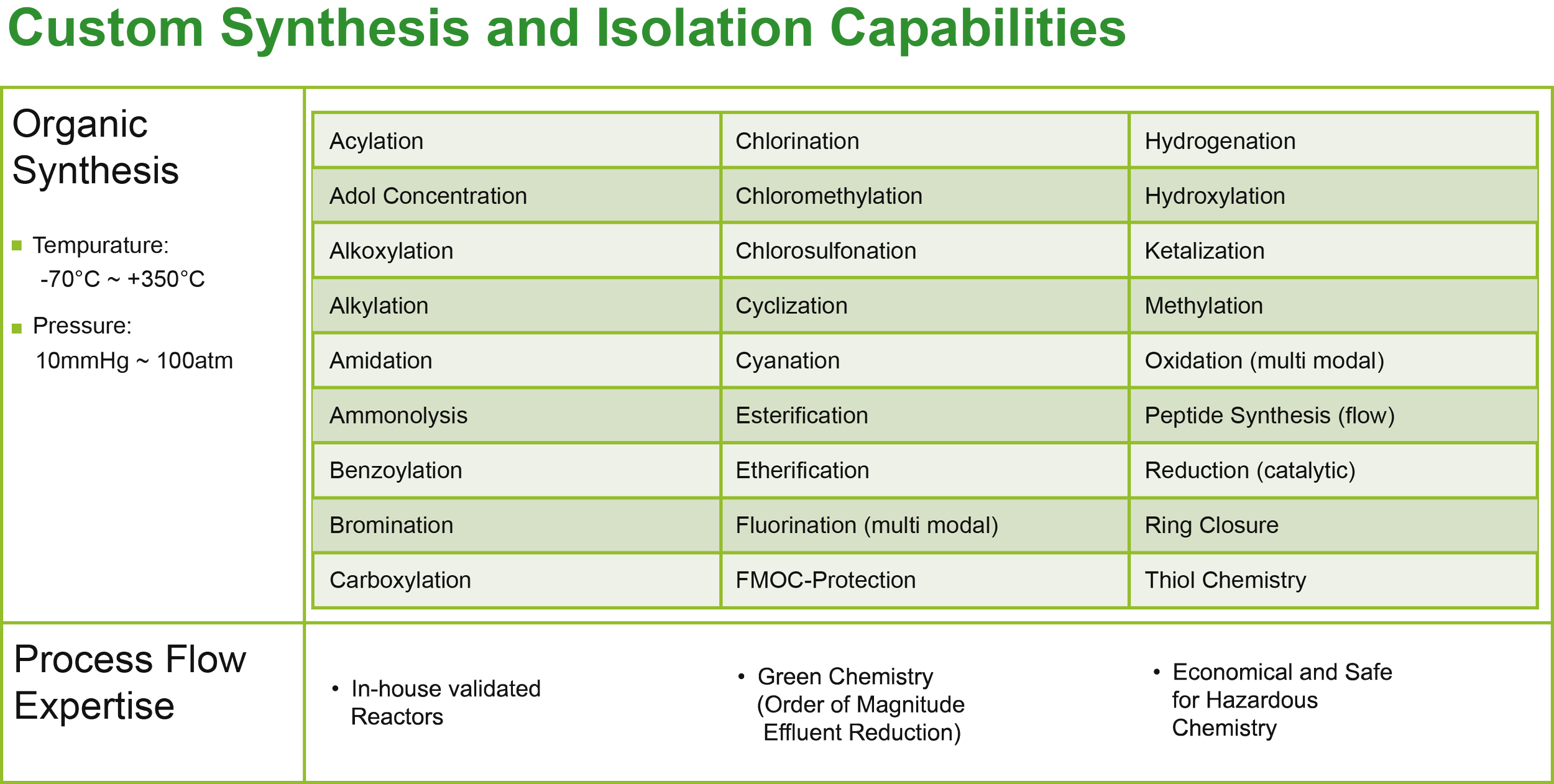
Custom Manufacturing
Built for the New Reality of Pharma Manufacturing
1. True Dual-Supply Capability
Not theoretical diversification — operational independence across China and India.
2. Technology-Led Manufacturing
Continuous flow, micro-reactors, and AI-enabled route optimization create structural performance advantages.
3. Backward Integration
Control of starting materials enhances reliability, cost stability, and supply security.
4. Speed Without Compromise
Faster reaction times and optimized synthesis routes accelerate development timelines.
5. Sustainability by Design
Lower energy consumption and reduced effluent generation support ESG priorities.
6. Safer Chemistry
Closed systems and improved process control reduce operator exposure and risk.
7. Trusted by Global Pharma
Decades of collaboration delivering advanced intermediates and regulatory starting materials.
A Partner for What’s Next
Whether supporting clinical programs or preparing for commercialization, we bring the technology, supply resilience, and execution capability required for modern pharmaceutical manufacturing.